


Antibody molecules have proven their value in clinical practice and are at the basis of many treatments. However, most of the easy targets are now already covered with monoclonal antibodies in clinical trials and the next generation of human monoclonal antibodies will have to target challenging molecules like integral membrane proteins and will require peculiar properties (agonist, antagonist, etc.) to open new avenues in patient treatment. The team has a long-standing expertise in protein engineering, particularly in the antibody field. We capitalize on this expertise for the development of original and innovative antibody-based molecules for in vitro and in vivo applications.
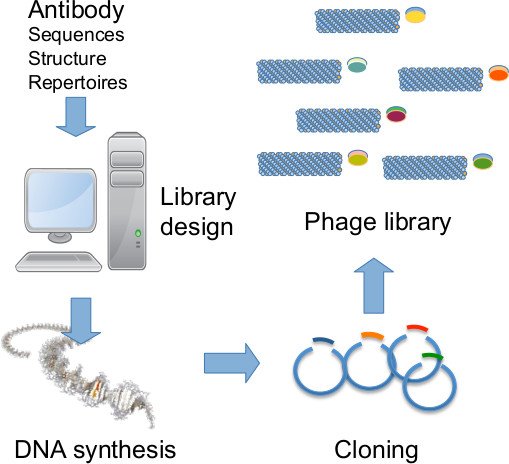
Aim 1. Synthetic library design and construction
Antibodies are now recognized as a key component of targeted therapies in cancer. The development of monoclonal antibodies has quickly evolved in the last 20 years towards a more robust and efficient pipeline. In particular, this has been accomplished through the development of display technologies (in particular phage display; G. Winter and G. Smith, 2018 Nobel Prize in Chemistry) and human libraries. We have made the choice 15 years ago to concentrate our work on the development of human fully synthetic antibody libraries because we think it allows a more efficient pipeline because of the high homogeneity of the designed libraries. The libraries developed by the team are the only academic libraries of human mAbs available for research in France. We continue to develop new innovative human synthetic libraries based on structure-function analyses of human repertoires with the aim of improving the functional diversity and the developability of the clones, together with an improved pipeline for the rapid and direct characterization of clones in IgG format.
Aim 2. Antibodies as biosensors and inhibitor
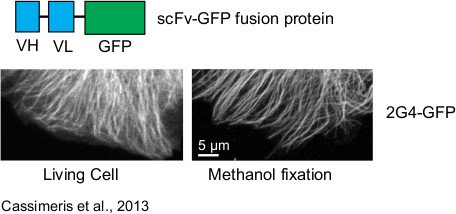
Antibodies are highly specific for their target and can be raised against virtually any part of a protein and in particular against large planar zones where protein interactions often take place and which are difficult to target with small organic molecules or peptides. This high specificity and affinity is routinely used in research laboratories and makes antibody the most widely used proteomic tool. Extending the use of antibodies in the cell is the main goal of the intrabody approach, which proposes to express antibody fragments within the cell to inhibit and modulate protein functions. However, natural antibodies are not adapted to the intracellular environment because of their large size and because the reducing environment of the cytoplasm prevents the formation of disulfide bridges and thus proper folding of antibodies. Our work on optimized human synthetic libraries (Aim 1) allows us to isolate antibody fragments with an exceptionally high stability and able to fold in the reducing environment that pertains in the cell cytoplasm. We currently focus on the use of intrabodies to target post-translational modifications in cells and their delivery in preclinical models.
Aim 3. Innovative antibodies for tumor targeting
Most of the antibodies used so far in clinic have been obtained by immunization. This has the disadvantage of restraining the potential diversity of the obtained molecules since it is almost impossible to raise antibodies against the epitopes conserved during evolution. In addition, in vitro display technologies allow to directly isolate fully human molecules and to develop specific selection pipelines for the identification of monoclonal antibodies with original binding properties. By combining our knowledge in synthetic antibody design and in vitro selection we currently aim at developing human antibodies, better adapted to the microenvironment of the tumor. Our main objective is to develop monoclonal antibodies with improved targeting efficiency even in the frequent case of expression of the targeted antigen in normal tissue; this will allow to optimize the efficacy/toxicity ratio to deliver improved monoclonal antibodies for human therapy.
Antibody molecules have proven their value in clinical practice and are at the basis of many treatments. However, most of the easy targets are now already covered with monoclonal antibodies in clinical trials and the next generation of human monoclonal antibodies will have to target challenging molecules like integral membrane proteins and will require peculiar properties (agonist, antagonist, etc.) to open new avenues in patient treatment. The team has a long-standing expertise in protein engineering, particularly in the antibody field. We capitalize on this expertise for the development of original and innovative antibody-based molecules for in vitro and in vivo applications.